Typical Flutter: the circuit, induction, and what truly confirms block
EP Master Series Flutter and Atrial Tachycardia — from mechanism to ablation
Typical flutter is one of the arrhythmias most familiar to every mapper.
And precisely because of that, it is also one of the most dangerous to oversimplify.
When a case looks obvious, the risk is stopping your thinking too early. The ECG looks familiar. The activation seems consistent. The CTI line appears straightforward. The tachycardia terminates. And without noticing it, the mapper replaces mechanism with habit.
But typical flutter is not just “a line case.”
Typical flutter is a macroreentrant atrial tachycardia, dependent on the cavotricuspid isthmus (CTI) as a critical segment of the circuit. In practical terms, this means a circuit that usually travels up the atrial septum, down the lateral wall of the right atrium, and crosses the CTI between the tricuspid annulus and the inferior vena cava. The ablation target is not the CTI by tradition. It is the CTI because of mechanism.
And that completely changes the logic of the case.
Because in typical flutter, the goal is not simply to terminate the arrhythmia. The goal is to demonstrate that the isthmus has truly stopped conducting in both directions. The endpoint remains bidirectional CTI block.
In this article, the goal is not to review typical flutter as a “classic” topic. The goal is to put the case back where it belongs: mechanism, maneuver, criteria, and confirmation.
Here is what you will find:
how to suspect typical flutter from the ECG without becoming overconfident
what defines typical flutter as macroreentry
how the mapper recognizes the circuit on intracardiac recordings
how to use entrainment to confirm CTI dependence
where the RF line is usually delivered and where the anatomical trap lies
how to truly confirm bidirectional block
and how to search for gaps when the line looks complete but still is not blocked
Figure 1
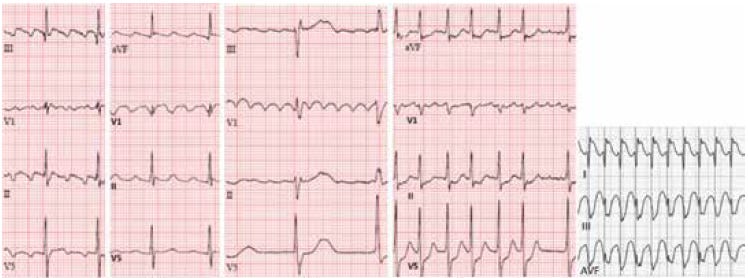
Comparisons between typical flutter, clockwise typical flutter, atypical flutter, and 1:1 flutter.
Source: Bonakdar HR. Atrial Flutter: Diagnosis and Management Strategies. In: Cardiac Arrhythmias. IntechOpen; 2018. DOI: 10.5772/intechopen.74850.
When the ECG makes you think of typical flutter
The ECG can raise a strong suspicion of typical flutter. But it does not establish the mechanism by itself.
In counterclockwise typical flutter, flutter waves are usually most visible in V1 and the inferior leads, with a sawtooth pattern, constant morphology, constant polarity, and no true isoelectric baseline between one wave and the next. In the classic counterclockwise form, septal activation makes the flutter waves predominantly negative in the inferior leads. In clockwise typical flutter, polarity may reverse, and V1 becomes even more important for interpretation. The chapter itself emphasizes that flutter-wave morphology can change in the presence of atrial disease, prior surgery, or previous ablation, which makes the ECG useful, but not definitive, for determining flutter type.
That is an important point for the mapper: the ECG suggests. It does not exempt you from testing.
In practical terms, I strongly suspect typical flutter when I find a regular atrial tachycardia, with a stable cycle length, continuous atrial activity, no true isoelectric interval, and repetitive morphology that is coherent with an organized right atrial reentry. But I only begin the case with that hypothesis. I do not finish the case there.
Figure 2
Caption: The classic ECG pattern helps a lot, but it does not replace mechanism.
Source: Bonakdar HR. Atrial Flutter: Diagnosis and Management Strategies. In: Cardiac Arrhythmias. IntechOpen; 2018. DOI: 10.5772/intechopen.74850.
Figure 3
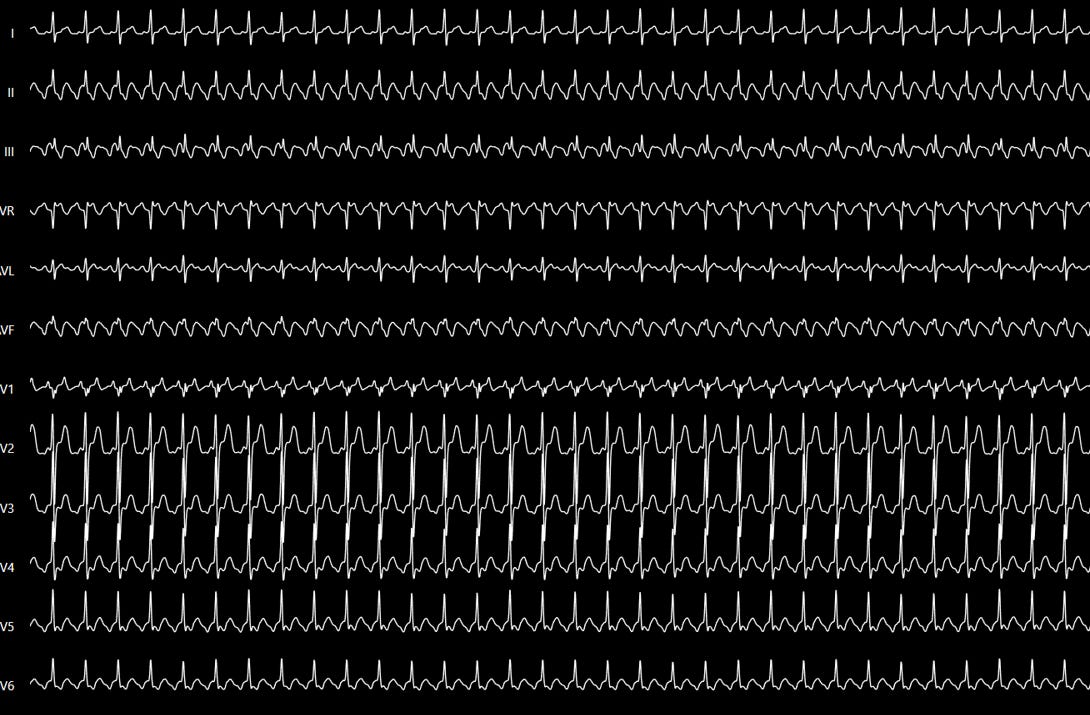
Caption: Typical flutter tends to occupy nearly the entire cycle. Focal AT usually does not.
Source: Bonakdar HR. Atrial Flutter: Diagnosis and Management Strategies. In: Cardiac Arrhythmias. IntechOpen; 2018. DOI: 10.5772/intechopen.74850.
Typical flutter is not a focus. It is a circuit.
This may be the most important conceptual shift in the entire article.
Typical flutter is a macroreentry. It is not a focal tachycardia arising from one point and spreading outward. It is a large, organized circuit, sustained by continuous propagation around anatomical and functional barriers, using the CTI as a critical conduction zone.
For the mapper, that changes everything.
If you think like a focal tachycardia, you search for the origin.
If you think like a macroreentry, you search for circuit coherence, activation direction, TCL coverage, and the critical conduction segment.
In other words:
In typical flutter, you are not chasing the earliest point.
You are dismantling a circuit.
What the intracardiac recordings need to show
In typical flutter, intracardiac electrograms show a constant cycle length, constant morphology, constant polarity, and an activation sequence that is consistent with macroreentry. In addition, endocardial recordings from the interatrial septum, upper anterolateral right atrium, and the CTI tend to cover 100% of the tachycardia cycle length. By contrast, in focal AT, even when looking only at the right atrium, activation usually covers less than 50% of the cycle.
This gives you an excellent discrimination point between a reentrant and a focal mechanism:
Does what I am see cover the whole cycle, as a large circuit should?
Or am I dealing with a more limited activation pattern, more consistent with a focal source?
In typical flutter, cycle-length stability is usually very high, with minimal variation. That also reinforces the impression of stable macroreentry rather than a more erratic or poorly defined mechanism.
Induction is not bureaucracy. It is hypothesis testing.
If the question of the case is whether the flutter is truly CTI-dependent, inducing the tachycardia is not just “making the arrhythmia appear.” It is creating the opportunity to test mechanism.
Distinguishing isthmus-dependent flutter from non-isthmus-dependent flutter is crucial before a curative ablation. Induction is often performed with programmed stimulation, frequently from the proximal CS, which tends to induce counterclockwise typical flutter. In some cases, isoproterenol may be needed to facilitate induction.
For the mapper, this means something very simple:
induction is there to confirm whether your circuit hypothesis truly holds up in practice.
Entrainment is not ornament. It is circuit confirmation.
The chapter is explicit: entrainment is the essential maneuver to confirm the diagnosis and determine whether the flutter is CTI-dependent. Overdrive pacing is performed at a cycle length 10–20 ms shorter than the tachycardia cycle length. During the maneuver, all atrial activity accelerates to the pacing rate; if pacing is progressively faster, progressive fusion may appear; and when pacing is stopped, the same tachycardia returns with the same cycle length and the same activation sequence.
In addition, check the morphological pattern on the diagnostic catheters during the tachycardia and compare it with what you see during entrainment pacing. If the site you are pacing changes the electrogram morphology, that already suggests the pacing site is probably not part of the circuit. If the morphology remains the same, that is an excellent sign that you are pacing inside the tachycardia circuit.
The key value we obtain from this is the post-pacing interval (PPI).
If the pacing site is in the circuit, or very close to it, the PPI will be very close to the tachycardia cycle length. When the catheter is at the CTI and the PPI is equal to, or within 20 ms of, the TCL, that supports the idea that the CTI is part of the circuit and confirms isthmus dependence.
If you suspect the CTI is part of the circuit, it is not enough to believe that based on how the case looks. Pace the CTI. Measure the PPI. See whether the response is truly that of a site belonging to the circuit.
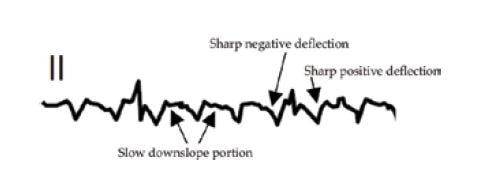
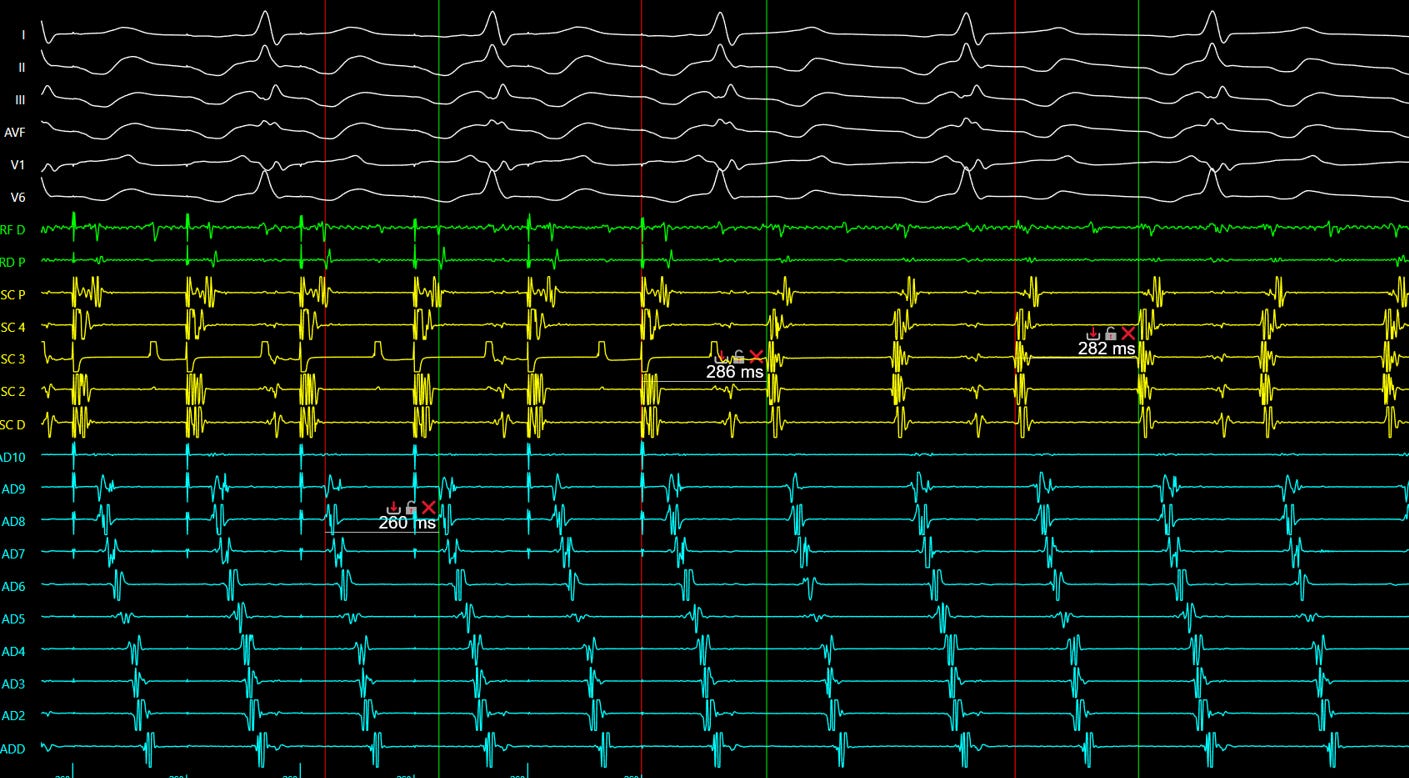
Figure 4
Caption: Entrainment. Return cycle of 286 ms minus tachycardia cycle length of 282 ms = 4 ms. Result: inside the circuit.
Source: Personal archive.
A practical entrainment framework in typical flutter
First, confirm a real and stable cycle length.
Then, with the mapping catheter at the CTI, perform overdrive pacing 10–20 ms faster than the tachycardia cycle.
Observe whether the tachycardia is being entrained coherently.
When pacing stops, measure the PPI at the same site.
If the PPI minus TCL is very short, especially up to 20 ms, that strongly favors the isthmus being part of the circuit.
The key sentence here is:
Entrainment is not only there to say, “this is flutter.”
It is there to test whether the point you are studying truly belongs to the circuit you are trying to interrupt.
That is why understanding entrainment is so important, not only in typical flutter, but in any reentrant tachycardia. The principle of entrainment is a principle of circuit localization.
Where the RF line is usually delivered
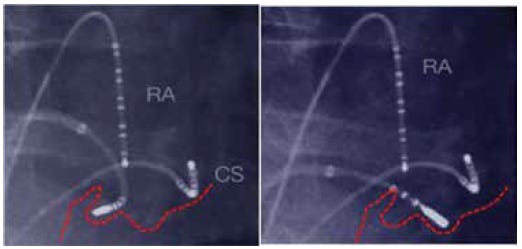
The classic line is delivered between the tricuspid annulus and the inferior vena cava, crossing the CTI.
The article describes a classic setup using three catheters: the ablation catheter, a CS catheter, and a duodecapolar catheter positioned around the tricuspid annulus. The duodecapolar catheter is placed so that its distal poles record the middle and lateral portions of the CTI. The ablation catheter is adjusted in LAO at approximately 6–7 o’clock. The first application is usually delivered near the tricuspid annulus, with a small A/V ratio. After each application lasting 30–60 seconds — depending on the catheter type, irrigated or 8 mm — the catheter is dragged back roughly 4 mm until a new sharp atrial electrogram is found, not far-field signal, and this is repeated until the border of the inferior vena cava is reached.
What the mapper needs to watch during the line
Adequate contact.
A true local electrogram.
An A/V ratio that helps orient position.
Coherent progression of the line.
And most importantly, the difference between an anatomically drawn line and a functionally blocked line.
Because those two things are not the same.
Where the anatomical difficulty lies
Not every CTI is the same.
The article describes variable anatomy: a wider lateral portion, a thinner central portion, the presence of concavity and pouches in a relevant proportion of patients, and the Eustachian ridge forming the posterior border of the isthmus. This anatomy can make line continuity much more difficult.
There is one particularly important detail for the article: the Eustachian ridge may prevent a simple drag-ablation technique from adequately reaching the most posterior part of the CTI. In these cases, it may be necessary to curl the catheter in order to access the floor of the pouch and complete the line.
In addition, the chapter reminds us that the septal/medial portion of the CTI lies close to the posterior extension of the AV node, which explains the higher risk of AV block when ablation is performed too medially, around 5 o’clock. The practical message is clear: the farther you stay from the septum, the lower the risk of that complication.
Figure 5
Caption: Sometimes the problem is not “lack of RF.” It is anatomy hiding the gap.
Source: Bonakdar HR. Atrial Flutter: Diagnosis and Management Strategies. In: Cardiac Arrhythmias. IntechOpen; 2018. DOI: 10.5772/intechopen.74850.
Terminating the tachycardia is not the endpoint
This point needs to be stated plainly.
Seeing flutter terminate during RF is important.
But it is not the endpoint.
The endpoint remains confirmation of bidirectional block, and the chapter itself emphasizes that this block should be checked at the end of the procedure and reassessed after 20–30 minutes, because conduction can recover.
That is the difference between a case that is executed and a case that is truly understood.
How the mapper confirms bidirectional block
The chapter organizes this well, and it can be turned into a field framework.
1. Activation sequence while pacing from one side of the line
When you pace from the medial side of the line, for example from the proximal CS, and a medial-to-lateral block is present, the activation wave must travel up the septum, around the right atrium, and only then reach the distal poles of the duodecapolar catheter. That is why the distal duodecapolar poles become the last ones to record activation.
2. Activation sequence in the opposite direction
To confirm lateral-to-medial block, you pace laterally to the line, for example from the distal poles of the duodecapolar catheter or with the ablation catheter at approximately 8 o’clock. If true block is present, activation travels up the lateral wall, around the superior right atrium, and then down the septum until it reaches the proximal CS.
This may be the simplest way to teach the logic to the mapper:
If block is present, the impulse is forced to go around.
If it still crosses directly, the line is not ready.
3. Trans-CTI conduction time
The chapter also gives a very useful number: a greater than 50% prolongation in conduction time across the CTI, or an absolute value of 150 ms or more, strongly favors isthmus block.
That is useful because it transforms the feeling of block into an objective check.
4. Differential pacing
This part is absolutely worth including because it gives real sophistication to the article.
To assess, for example, lateral-to-medial block, you first pace close to the line, such as from duodecapolar 1–2, measure the time to the proximal CS, and then pace farther from the line, such as from duodecapolar 3–4. If block is present, that interval becomes shorter as you move away from the line. If the CTI is still conducting, the opposite happens: the interval becomes longer, because the wavefront is still crossing the isthmus directly.
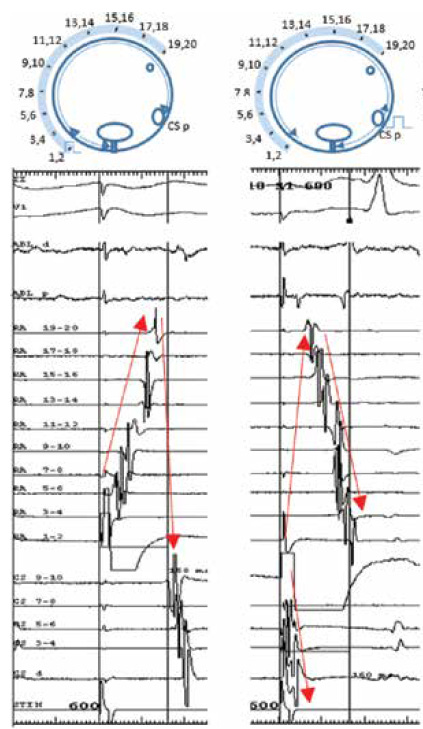
Figure 6
Caption: Bidirectional block is not a visual impression. It is activation behavior tested in both directions.
Source: Bonakdar HR. Atrial Flutter: Diagnosis and Management Strategies. In: Cardiac Arrhythmias. IntechOpen; 2018. DOI: 10.5772/intechopen.74850.
When the line looks complete, but a gap is still there
This is one of the most valuable parts of the chapter.
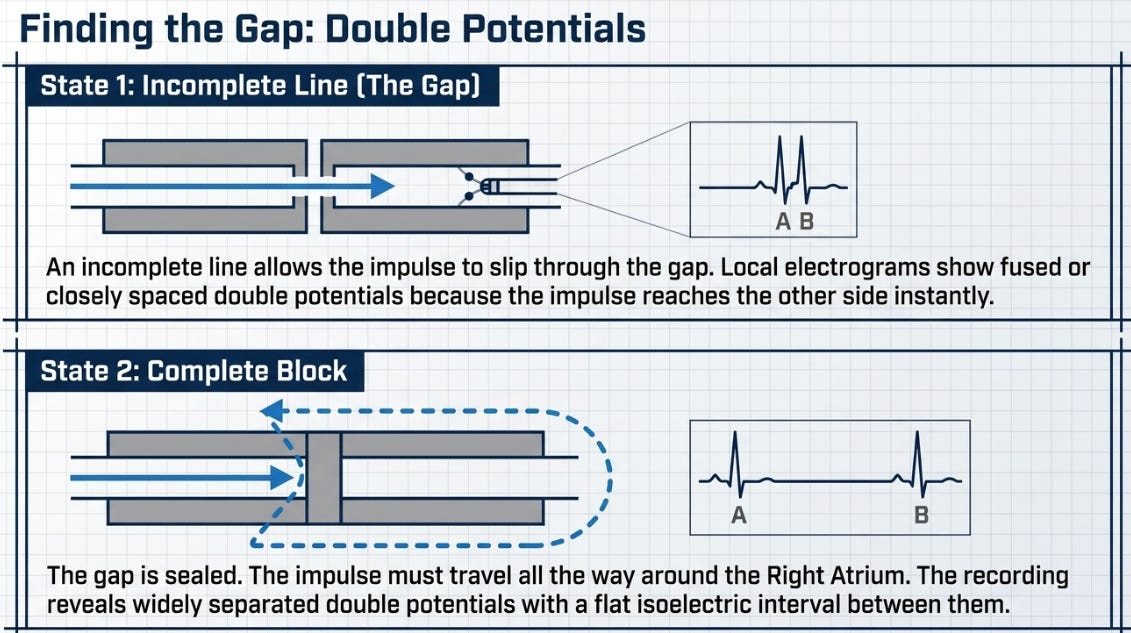
The text reminds us that residual gaps are common because of isthmus anatomy and pouches, and that finding them is mandatory to achieve complete bidirectional block and reduce recurrence. These gaps can be detected through fractionated electrograms and through the behavior of double potentials along the line. When you move the catheter toward the gap, the interval between the two components of the double potential becomes progressively shorter, until, at the gap itself, the signals fuse. When the line is truly blocked, the interval between the components becomes longer again because the wavefront now needs to travel all the way around the right atrium to reach the other side of the line.
This deserves to become a signature sentence:
A gap is tracked by signal physiology.
Figure 7
Caption: The gap is not where the line looks bad. It is where physiology shortens the interval and fuses the signals.
The mapper’s framework for calling true block
If I had to summarize all of this into one practical field logic, it would be this:
After the CTI line, I only call true block when I find a coherent set of signs.
The tachycardia terminated, but I do not stop the case there.
I test activation from one side of the line and see whether the impulse is now forced to travel around the atrium.
I test the opposite direction and look for the same logic.
I compare trans-CTI time with baseline and want to see important prolongation, ideally more than 50% or at least around 150 ms.
I use differential pacing when I need to distinguish true block from residual conduction.
I walk the line searching for gaps whenever something still does not fit.
That is how the mapper stops operating by impression and starts operating by electrophysiological evidence.
What typical flutter teaches about the mapper
Perhaps the greatest educational value of typical flutter is not that it is common.
Perhaps it is that it exposes, very clearly, the difference between execution and understanding.
The mature mapper thinks:
“Did I demonstrate true bidirectional block?”
That is the right question.
In the end, typical flutter is not simple because the circuit is simple. It looks simple because we have seen it so many times. But in electrophysiology, repetition without interpretation creates too much confidence and not enough precision.
The goal is not simply to cross the CTI with RF.
The goal is to demonstrate, with criteria, that the circuit has lost its critical conduction pathway.
And that difference completely changes the level of the case.
References
Bonakdar HR. Atrial Flutter: Diagnosis and Management Strategies. IntechOpen. 2018. DOI: 10.5772/intechopen.74850.
Saoudi N, Cosío F, Waldo A, et al. A classification of atrial flutter and regular atrial tachycardia according to electrophysiological mechanism and anatomical bases.
Chen J, De Chillou C, Basiouny T, et al. Cavotricuspid isthmus mapping to assess bidirectional block during common atrial flutter radiofrequency ablation.
Oral H, Sticherling C, Tada H, et al. Role of transisthmus intervals in predicting bidirectional block after ablation of typical atrial flutter.
Shah D, Haïssaguerre M, Takahashi A, et al. Differential pacing for distinguishing block from persistent conduction through an ablation line.
EP Master
Thank you for reading EP Master. Subscribe to receive new content.